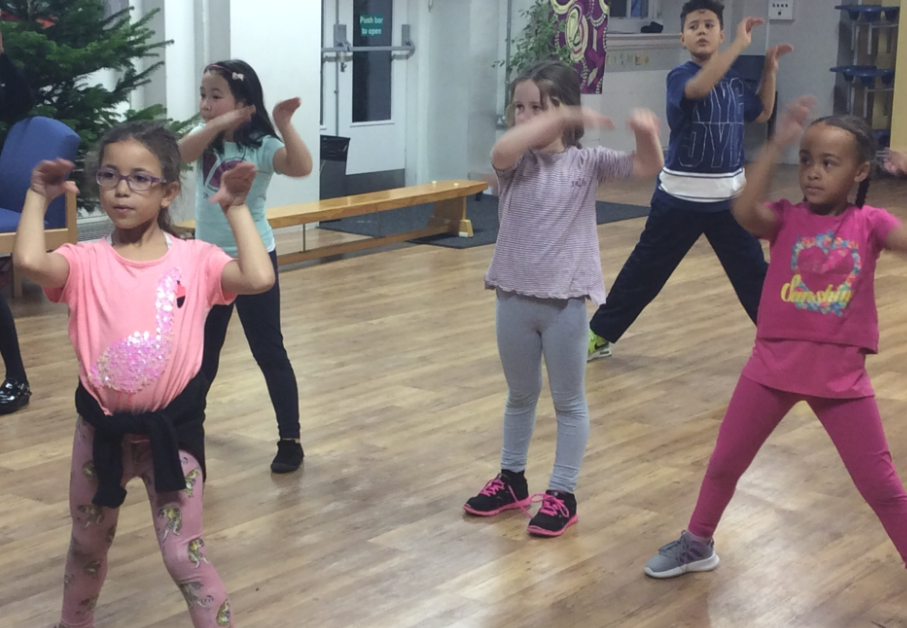Primary Phase Clubs
The activities offered at Trinity Primary range from Zumba to Forest Schools to Embroidery. A breadth of creative and active options are available for the pupils in order to unearth their passions and interests outside the classroom.
If you have any questions about clubs please let Miss Apsey, our onsite provision lead, know.
Here are a sample of the clubs we have run:
 Book club – Mrs Fielder
Book club – Mrs Fielder
This club will be run for children across the school to encourage a love of reading. Working in small groups, the children will develop their fluency and stamina for reading while creating character profiles and sharing their thoughts on the stories.
Art – Ms Coventry
At lunch time, children will work with a specialist art teacher to create an item by the end of the half term using a variety of skills and tools. The pupils will use a variety of methods to design and produce works of art in different media.
Embroidery – Miss Lewis
After practising the basics of cross stitch for the initial weeks, the pupils will build up their patterns to create seasonal images. They will be able to choose their own design and be given a range of colourful threads to create a unique product.
Choir – Mrs Bibby-Thompson
With great direction the high quality of performance by the Trinity Choir has been maintained and is recognised for its positive role in our school community.
Multi Skills – Fit For Sport
 By providing a wide variety of games, this is a fun, challenging and enjoyable starting point for children’s involvement in any future physical activities that the child may subsequently choose to participate in.
By providing a wide variety of games, this is a fun, challenging and enjoyable starting point for children’s involvement in any future physical activities that the child may subsequently choose to participate in.
For more information please follow the link,
https://www.fitforsport.co.uk/
Football – Premier Sports
A popular club for boys and girls of all abilities. This club will guide children to undertake a series of footballing activities in a structured and fun way.
For more information please follow the link,
https://www.premier-education.com/
Zumba – Zumba Stars London
Zumba fuses fitness and entertainment. The classes blend upbeat world rhythms with easy-to-follow choreography for a total-body workout. You do not need rhythm, co-ordination or dance ability - all you need is to shake your body to the music and have FUN!
For more information please follow the link
https://www.zumbastarslondon.co.uk/
Yoga – Yoga Bugs

For more information please follow the link
Karate – SKFK
The club allows for a combination of guidance and enjoyment. Students can take part for fun or as the opportunity to progress in belt grading and learning further in the art of karate.
For more information please follow the link
Forest School – Forries Education
The benefits of getting our children outdoors - having adventures, taking responsibility, learning how
For more information please follow the link